Baran Lab
@BaranLabReads
Followers
23,315
Following
121
Media
288
Statuses
1,410
Electrifying chemistry...
La Jolla, CA
Joined January 2014
Don't wanna be here?
Send us removal request.
Explore trending content on Musk Viewer
Liam Payne
• 1373821 Tweets
Liam Payne
• 1373821 Tweets
Argentina
• 575024 Tweets
Bret
• 352401 Tweets
hobi
• 327693 Tweets
hoseok
• 296087 Tweets
jhope
• 239564 Tweets
García Luna
• 213563 Tweets
WELCOME BACK J-HOPE
• 204535 Tweets
Fox News
• 200463 Tweets
Bear
• 186337 Tweets
ブロック
• 166195 Tweets
Lo de Liam
• 163680 Tweets
maya
• 150937 Tweets
MSNBC
• 120529 Tweets
Calderón
• 118425 Tweets
Snapchat
• 110608 Tweets
Buenos Aires
• 102705 Tweets
Directioner
• 84402 Tweets
Palermo
• 74046 Tweets
Muñoz
• 73641 Tweets
DE LA MANO DEL SEÑOR
• 59768 Tweets
Meech
• 52680 Tweets
Dove
• 30868 Tweets
Bluesky
• 30196 Tweets
スーパームーン
• 27174 Tweets
Hannah
• 26129 Tweets
story of my life
• 23531 Tweets
#OneDirection
• 17750 Tweets
Ambush
• 17634 Tweets
#AEWDynamite
• 10800 Tweets
イーロン
• 10583 Tweets
Last Seen Profiles
A message to the
@scrippsresearch
chemistry community from
@DonnaBlackmond
and Phil regarding the
@angew_chem
essay posted here for anyone interested on Openflask:
23
288
868
Can total synthesis provide a viable blueprint for a med. chem. exploration of Taxol in the absence of semi-synthesis? Like proving an obscure mathematical theorem, thats the fundamental q. we set out to answer >13 years ago. Appearing now on
@ChemRxiv
:
23
176
640
Presenting an electrifying C–C bond forming method that can drastically shorten the routes to a myriad of structures, appearing today in
@ChemRxiv
().
9
83
558
Today we report in
@ChemRxiv
a scalable solution to the total synthesis (and structural reassignment) of the Portimines (). In collaboration with
@_chrisgparker_
and Luke Lairson the remarkable biological activity of this natural product family was revealed
12
78
479
TAGETITOXIN! With only one of its 11 carbons without a heteroatom attached, its probably the most polar organic compound we have ever made... A total synthesis, Just Accepted
@J_A_C_S
: Behind-the-scenes:
7
68
429
Degrade proteins, not your work schedule! A new method appearing today
@ChemRxiv
() simplifies access to molecular glues (like PROTACS) to save medicinal chemists valuable time. One-step access from cheap commercial materials, a trivial experimental setup,
5
68
411
The 2021 edition of Heterocycles
@scrippsresearch
is coming in a few weeks. New Zoom format open to the world. Contact mpalkowitz
@scripps
.edu for link. Syllabus coming soon.
6
98
389
14 simplified natural product syntheses reported today in
@ChemRxiv
: . These syntheses are enabled by a next generation doubly decarboxylative cross coupling (dDCC) that tolerates alpha-heteroatom substituents. In addition to the
2
66
372
Bye Bye Grignards (at least for ketone synthesis 😉). A radical alternative to anionic chemistry, out now in
@J_A_C_S
:
7
92
352
Today in
@J_A_C_S
a solution to a longstanding challenge in the field: Serine Selective Bioconjugation (). In collaboration with
@gbernardes_chem
,
@EastgateMartin
,
@Dawson_Group
labs.
6
60
343
The final version of this study was published today in
@Nature
:
Today we report in
@ChemRxiv
a scalable solution to the total synthesis (and structural reassignment) of the Portimines (). In collaboration with
@_chrisgparker_
and Luke Lairson the remarkable biological activity of this natural product family was revealed
12
78
479
6
62
341
Today in
@ChemRxiv
() we present an unusually efficient solution (4 years in the making) to a classic challenge in terpene synthesis enabled by electrocatalysis (Ag-nanoparticle electrodes!) and radical retrosynthesis.
5
47
328
Anomeric Amides to the rescue! Halogenate even the most stubborn systems with a new class of reagents. Appearing today in
@ChemRxiv
- a fun collaboration with Abbvie, BMS and Pfizer.
4
48
323
Got Nitrogen? A concise enantioselective synthesis of the marine-derived alkaloid KB343 is disclosed today in
@ChemRxiv
: .
9
45
320
Appearing today in
@J_A_C_S
: The first enantioselective doubly decarboxylative cross couplings (). A unique approach to chiral alpha-substituted carbonyls. The method, combined with radical retrosynthetic logic, can dramatically simplify synthesis.
6
36
316
Simple, practical, and broadly applicable. Next generation decarboxylative arylation, appearing today in
@ChemRxiv
:
6
40
308
It was only a matter of time: Phosphorus(V) gets "radicalized".
Appearing today in
@ChemRxiv
, access to new chemical space through the first stereocontrolled radical reactions of P(V): . Another awesome collaboration with the team at BMS.
4
48
300
ONE (aziridine) RING TO RULE THEM ALL! The total synthesis of Dynobactin A, appearing today in
@ChemRxiv
:
4
48
302
Simple, Kg-Scale Electrochemical Decarboxylative Olefination appearing today in
@ChemRxiv
() with Abbvie (
@KaidHarper
) facilitated by Alternating Polarity...
6
41
299
Modern Alchemy with rAP. Appearing today in
@ChemRxiv
is a solution to the longstanding (>100 year) challenge of extending the scope of the original Kolbe reaction beyond simple hydrocarbons bearing no functional groups:
5
36
277
A useful guide to decarboxylative cross coupling, especially for medicinal chemists, online today
@ChemRxiv
:
1
46
280
⚡️An electrifying total synthesis to start 2024⚡️! The total synthesis of Dragocins A-C appearing today in
@ChemRxiv
: . Happy New Year! 🥂
5
31
272
Radical retrosynthesis strikes again. Primary RAEs can be mixed and matched with tertiary RAEs or olefins to make quaternary centers with a comical level of simplicity. Check out these applications. A fun collaboration with the great and powerful
@Shenvi_Lab
:
2
41
270
The final version of this work was published in
@NatureChemistry
:
Anomeric Amides to the rescue! Halogenate even the most stubborn systems with a new class of reagents. Appearing today in
@ChemRxiv
- a fun collaboration with Abbvie, BMS and Pfizer.
4
48
323
1
36
265
Published today in
@ScienceMagazine
:
Radical retrosynthesis strikes again. Primary RAEs can be mixed and matched with tertiary RAEs or olefins to make quaternary centers with a comical level of simplicity. Check out these applications. A fun collaboration with the great and powerful
@Shenvi_Lab
:
2
41
270
3
29
257
Today we report
@ChemRxiv
a practical method to obtain C–4 alkylated pyridines with exquisite site-selectivity in collaboration with
@GodineauEdouard
of
@Syngenta
using simple Minisci decarboxylation:
9
35
255
The lab was honored to play a very tiny role in this story with
#Pfizerchemistry
as radical-based decarboxylative amino acid synthesis was used to optimize Paxlovid.
2
33
249
A Simple Method to Couple UNACTIVATED olefins and ketones enabled by e-chem when purely chemical approaches fail. A fun collaboration with
@MinteerLab
,
@PGE13
of Minakem, and
@asymchem
as part of the
@NSF_CSOE
. Appearing today on
@ChemRxiv
:
2
32
241
MEDICINAL CHEMISTS: Looking to increase your Fsp3? Synthesize in 3D just as you have been doing in 2D for eons by combining biocatalytic C–H oxidation and radical retrosynthesis (enabled by electrocatalysis). Appearing today in
@ChemRxiv
: . A fantastic
4
32
241
A radically electrified approach to the total synthesis of Vitamin D appearing today in
@ChemRxiv
(). Thanks to
@LEOPharmaUS
for another great collaboration.
5
30
239
"Dump, stir, electrify" - Quaternary centers made easily through decarboxylative arylation of tertiary acids with (hetero)aryl bromides. Appearing today in
@ChemRxiv
: . A super fun collaboration with BMS, Biogen, LEO Pharma, and Enamine.
2
30
235
JAWSamycin accessed in a unique modular way enabled by e-borylation appearing today in
@ChemRxiv
: . Method development, mechanistic analysis (with
@DonnaBlackmond
), scope, and applications, led by
@lmbarton93
.
5
44
233
CYCLOPAMINE! A total synthesis with LEO Pharma collaborators. Appearing today in
@ChemRxiv
:
8
37
234
Today in
@ChemRxiv
(): Democratized access to stereo pure thioisosteres of life's building blocks with promising translational potential in medicine. New reagents and a completely different strategy coupled to some amazing collaborators enabled this study.
2
39
231
Today in
@ChemRxiv
() a collaborative study on a combinatorial approach to oxidizing sp3-C–H bonds. N-ammonium ylide-based oxidants: Developed based on first principles, guided by computation, and activated electrochemically. Modular, selective, simple.
1
38
228
Simple access to Aryl-SO2R bonds using aryl halides,SO2, Ni, and some electrons. Appearing today in
@ChemRxiv
:
4
27
226
A fun collaboration to simplify amino acid synthesis through radical retrosynthesis.
Check out our latest application of Ag/Ni electrocatalysis to the synthesis of unnatural amino acids from
@BaranLabReads
! Great job
@g_laudadio
and
@pneigenfind98
working with the amazing
@chemveda
team and
@biogen
collaborators!⚡️
0
4
68
2
30
222
Enantioselective preparation of trivial looking dialkyl carbinols has historically relied on two disconnections based on polar bond analysis resulting in low ideality. Today in
@ChemRxiv
we show how radical retrosynthesis can unlock simple access:
4
19
212
Thanks to
@ChemScrapes
for his generosity in donating his amazing talent for the cover art of this paper. It sums up the whole story pretty well 😄BTW, You can order his awesome book here:
Our taxane perspective from
@BaranLabReads
is featured on the front cover of
@JOC_OL
. Our work is truly complete with this terrific and humorous art created by
@ChemScrapes
. Everyone should check out his work/book right now!
0
1
39
5
42
217
Short on time but need an aryl-alkyl bond?
@mx_million
and
@g_laudadio
give a video guide to reaction setup.
1
23
218
A classic reaction for C–C bond formation, NHK, electrified, appearing today in
@ChemRxiv
: . An exciting collaboration with
@DonnaBlackmond
and
@sarah_reisman
groups to uncover its enhanced scope, real-world utility, and intricate mechanism.
3
29
216
Radical retrosynthesis enables LEGO-like natural product assembly, exemplified with 14 more natural product syntheses appearing today in
@Nature
:
14 simplified natural product syntheses reported today in
@ChemRxiv
: . These syntheses are enabled by a next generation doubly decarboxylative cross coupling (dDCC) that tolerates alpha-heteroatom substituents. In addition to the
2
66
372
2
33
216
Modern alchemy appearing today in
@ScienceMagazine
:
Commentary (thank you Prof. Ye!):
Modern Alchemy with rAP. Appearing today in
@ChemRxiv
is a solution to the longstanding (>100 year) challenge of extending the scope of the original Kolbe reaction beyond simple hydrocarbons bearing no functional groups:
5
36
277
5
37
214
The final version of this study has appeared today in
@NatureChemistry
:
Today in
@ChemRxiv
(): Democratized access to stereo pure thioisosteres of life's building blocks with promising translational potential in medicine. New reagents and a completely different strategy coupled to some amazing collaborators enabled this study.
2
39
231
6
37
209
Chiral amino alcohols without the hassle, radically simplified appearing today in
@ChemRxiv
:
3
27
208
FAREWELL WILLIAMSON ether synthesis: An approach to hindered ethers that capitalizes on e-generated cations. Deposited
@ChemRxiv
here:
5
62
196
This study was published today in
@angew_chem
:
Alpha-Amino ketones radically simplified (ROCl + RAE) under enantioselective Ni-catalysis, appearing today in
@ChemRxiv
with
@YangTotSyn
:
3
18
163
2
19
205
Electrocatalytic Enantioselective Alkyl-NHK reactions, appearing today in
@ChemRxiv
: . A fun
@NSF_CSOE
collaboration with the amazing
@sarah_reisman
,
@Sigman_Lab
, and
@MinteerLab
0
33
200
A MAXimally rewarding total synthesis of MAXimicin has just appeared in
@J_A_C_S
: . For MAXimum details, check out Kyle’s behind-the-scenes account at Open Flask:
6
29
198
Final version of this work has appeared in
@angew_chem
:
Simple, Kg-Scale Electrochemical Decarboxylative Olefination appearing today in
@ChemRxiv
() with Abbvie (
@KaidHarper
) facilitated by Alternating Polarity...
6
41
299
3
29
199
The final version of this highly collaborative work
@NSF_CSOE
has appeared today in
@Nature
: -- Check it out as there is a ton of new data that was added since last year!
3
36
196
The final version of this work was published today in
@J_A_C_S
:
Electrocatalytic Enantioselective Alkyl-NHK reactions, appearing today in
@ChemRxiv
: . A fun
@NSF_CSOE
collaboration with the amazing
@sarah_reisman
,
@Sigman_Lab
, and
@MinteerLab
0
33
200
1
10
193
A SILVER BULLET FOR TERPENE SYNTHESIS, published today in
@ScienceMagazine
: - Behind the scenes at Openflask: - Phil doing the reaction:
Today in
@ChemRxiv
() we present an unusually efficient solution (4 years in the making) to a classic challenge in terpene synthesis enabled by electrocatalysis (Ag-nanoparticle electrodes!) and radical retrosynthesis.
5
47
328
5
27
192
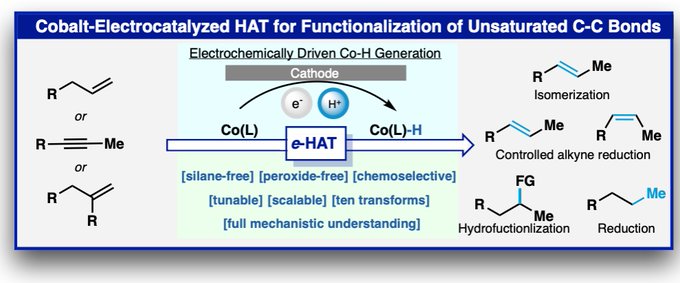
Anomeric Nitroamide Enabled, Cobalt Catalyzed Alkene Hydronitration: Appearing today in
@ChemRxiv
:
Tertiary nitroalkanes, as well as its reduced products, α-tertiary amines, play an essential role in drug discovery either as key synthetic precursors or
2
31
192
This work has now appeared in
@J_A_C_S
, with loads of color, all for free:
A Simple Method to Couple UNACTIVATED olefins and ketones enabled by e-chem when purely chemical approaches fail. A fun collaboration with
@MinteerLab
,
@PGE13
of Minakem, and
@asymchem
as part of the
@NSF_CSOE
. Appearing today on
@ChemRxiv
:
2
32
241
1
13
184
Now available in
@ChemRxiv
, a guide (written with
@YuKawamata
) to a useful new technique for synthesis: rapid alternating polarity (rAP). .
1
29
184
Published today in
@angew_chem
:
Degrade proteins, not your work schedule! A new method appearing today
@ChemRxiv
() simplifies access to molecular glues (like PROTACS) to save medicinal chemists valuable time. One-step access from cheap commercial materials, a trivial experimental setup,
5
68
411
2
22
185
An interesting perspective on emerging ways to desaturate carbonyl groups, one of the most basic organic reactions, appearing today in
@ACSCatalysis
: . Written with our collaborators at
@Minafin_Group
,
@PGE13
and
@FabSerpier
.
3
37
178
The updated version of this work has appeared today
@ChemRxiv
now with applications, scale-up, and mechanistic studies.
"Dump, stir, electrify" - Quaternary centers made easily through decarboxylative arylation of tertiary acids with (hetero)aryl bromides. Appearing today in
@ChemRxiv
: . A super fun collaboration with BMS, Biogen, LEO Pharma, and Enamine.
2
30
235
1
21
177
C to B in 4 minutes: A next generation decarboxylative borylation using Cu catalysis
#realtimechemistry
2
37
172
The final version of this work has now appeared in
@J_A_C_S
: Read
@yuzururururu
's post at Open-Flask for a behind-the-scenes account:
Can total synthesis provide a viable blueprint for a med. chem. exploration of Taxol in the absence of semi-synthesis? Like proving an obscure mathematical theorem, thats the fundamental q. we set out to answer >13 years ago. Appearing now on
@ChemRxiv
:
23
176
640
2
20
168
The final version of this study has appeared today in
@J_A_C_S
:
Today we report
@ChemRxiv
a practical method to obtain C–4 alkylated pyridines with exquisite site-selectivity in collaboration with
@GodineauEdouard
of
@Syngenta
using simple Minisci decarboxylation:
9
35
255
2
23
171
Curious how oligonucleotides are made on scale and what the future holds? Check out our review with
@LovelockLab
appearing today in
@ScienceMagazine
:
0
26
172
The final version of this work has appeared today in
@angew_chem
:
The updated version of this work has appeared today
@ChemRxiv
now with applications, scale-up, and mechanistic studies.
1
21
177
1
28
169
The final version of this work has just appeared in
@J_A_C_S
:
It was only a matter of time: Phosphorus(V) gets "radicalized".
Appearing today in
@ChemRxiv
, access to new chemical space through the first stereocontrolled radical reactions of P(V): . Another awesome collaboration with the team at BMS.
4
48
300
2
17
170
Electrochemical Cyclobutane Synthesis in-flow: Another great collaborative study with
@JJSabatini
now appearing in
@OPRD_ACS
: (special thanks to for flow equipment!)
1
24
169
Published today in
@JOC_OL
:
0
18
167
Need a phosphate? A useful method to phosphorylate alcohols using a Ψ-reagent appearing today in
@ChemRxiv
: . Another great collaboration with
@EastgateMartin
and the BMS team.
2
30
164